
February 16, 2026
Data Lineage in Pharmacovigilance: Proving Where Every Data Point Came From
Pharmacovigilance inspections no longer focus only on whether data exists. They focus on whether data can explain itself. Inspectors ask how a case was captured, how it moved across systems, who modified it, and what controls governed each step. When these answers rely on memory rather than evidence, findings follow.
This expectation has elevated data lineage from a technical concept to a regulatory requirement. In modern pharmacovigilance, proving where every data point came from is essential to demonstrating control, integrity, and compliance.
What Data Lineage Means in Pharmacovigilance?
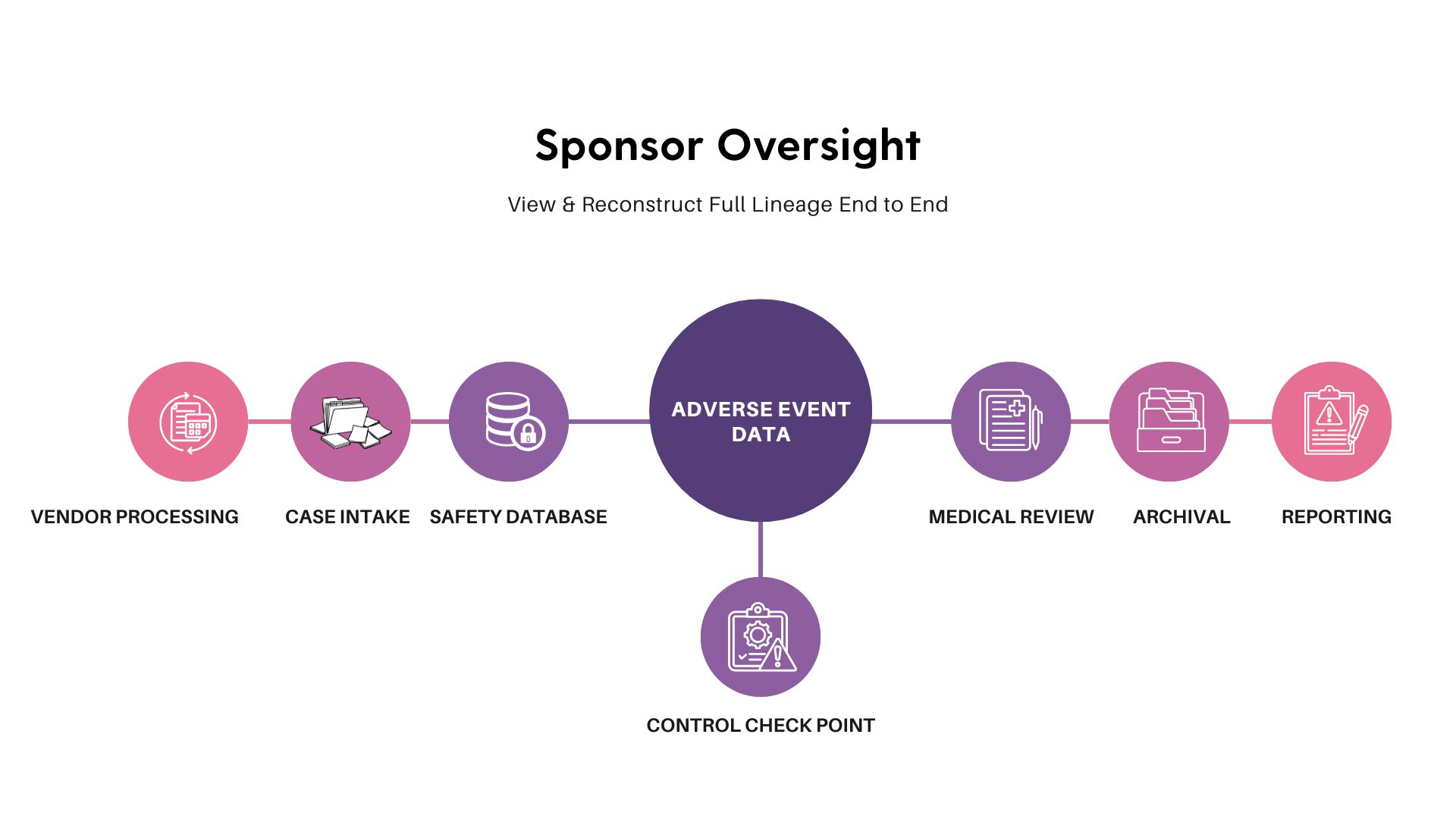
Data lineage is the documented, traceable history of safety data from origin to submission. It shows how information was captured, transformed, validated, transferred, and reported across systems and partners.
In pharmacovigilance, lineage applies to adverse event cases, follow-up data, coding decisions, seriousness assessments, and regulatory submissions. It connects source documents, safety databases, analytics tools, and reporting gateways into a single, explainable chain.
Lineage is not merely an audit trail. Audit trails record actions. Lineage explains flow, dependency, and control.
Why Regulators Care About Lineage?
Regulators evaluate lineage to assess data integrity. They want assurance that safety decisions are based on information that has not been altered, lost, or misinterpreted during movement.
Authorities such as the European Medicines Agency and the U.S. Food and Drug Administration increasingly test whether sponsors can reconstruct the path of a single data point across systems. If lineage cannot be demonstrated clearly, confidence in the entire dataset is weakened.
Guidance from the International Council for Harmonisation emphasises traceability and reproducibility across the product lifecycle. Without lineage, these principles cannot be proven.
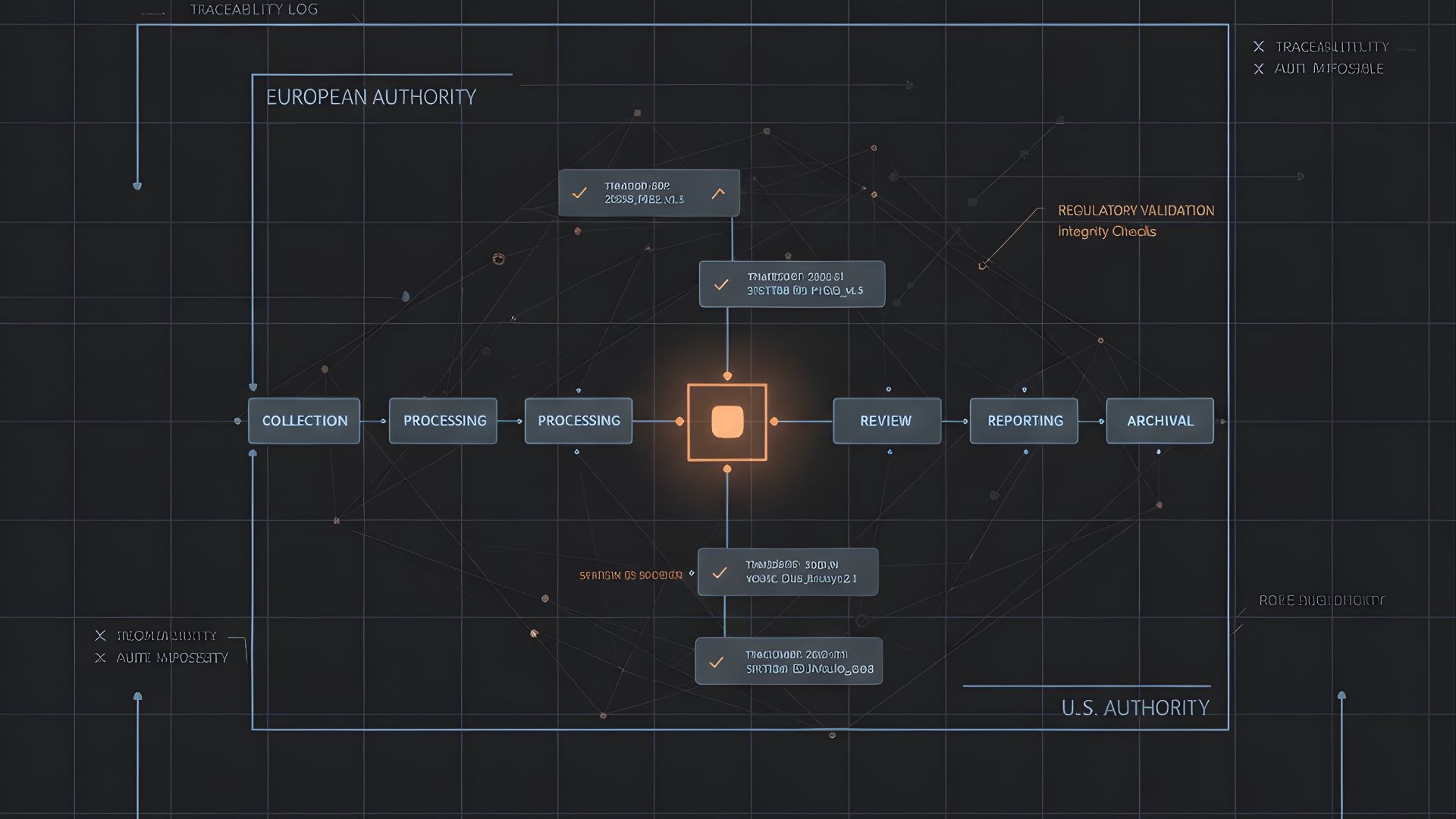
Where Data Lineage Commonly Breaks?
Lineage gaps most often appear during system transitions. Data migration from legacy databases introduces transformation logic that is not always fully documented. When field mappings, reconciliation outcomes, or validation checks cannot be reproduced, lineage is interrupted.
Another common break occurs in vendor handoffs. When different partners perform case processing, medical review, or reporting, data may move across platforms without a unified oversight layer. Each system may have its own audit trail, but the end-to-end narrative is lost.
Manual interventions also create risk. Offline edits, spreadsheet reconciliations, or email-based corrections may resolve immediate issues but leave no controlled record within the PV environment. During inspection, these gaps become critical.
The Inspection Impact of Weak Lineage
When lineage is unclear, inspectors broaden their scope. Questions expand from individual cases to system governance, change control, and vendor oversight. What began as a data query becomes a systemic review.
Weak lineage does not always result in isolated findings. It often leads to observations about loss of control, inadequate validation, or insufficient oversight. These outcomes carry higher remediation costs and longer regulatory impact.
Designing Lineage Into PV-IT Systems
At Fidelity Health Services, data lineage is treated as a design requirement rather than an afterthought. PV-IT environments are structured so that data flow is inherent, visible, and reconstructable.
Each data transfer is governed by validated interfaces. Transformation rules are documented and approved. Reconciliation reports confirm completeness and accuracy. Changes are managed through controlled workflows that preserve historical context.
By integrating safety databases, analytics platforms, and reporting gateways within a unified architecture, lineage remains intact even as systems evolve.
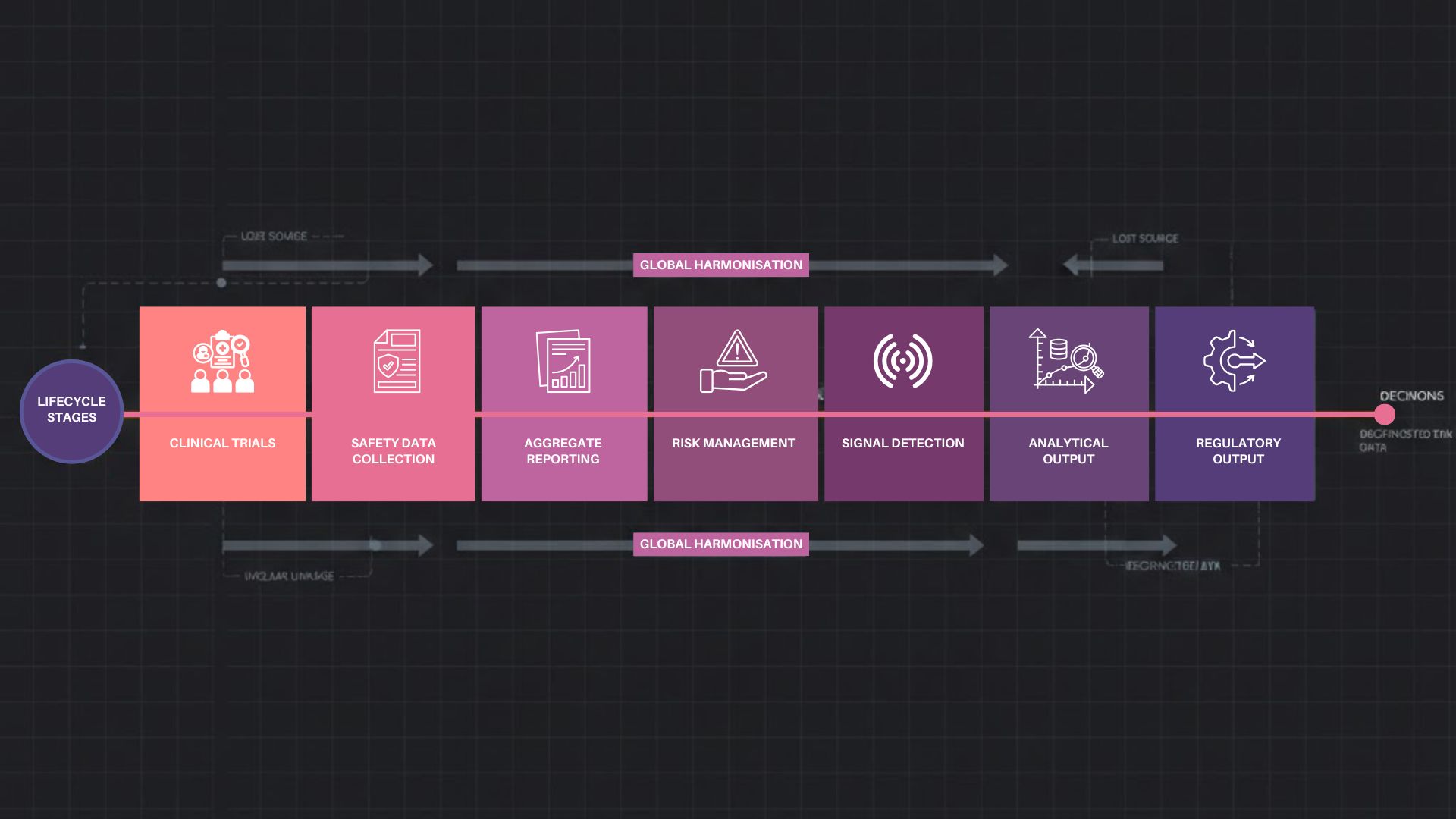
Lineage Across the Product Lifecycle
Data lineage is not limited to post-marketing surveillance. It begins in clinical development and extends through commercialisation and lifecycle management.
Clinical safety data feeds aggregate reports, risk management plans, and signal detection activities. Without lineage, linking outcomes back to source data becomes difficult. With lineage, organisations can demonstrate consistency across submissions and decisions.
The World Health Organisation promotes harmonised data exchange precisely because lineage enables global signal interpretation and trust.
Lineage as a Preventive Control
Strong lineage reduces inspection risk before it appears. When systems can explain how data moved and why changes occurred, inspectors spend less time probing governance and more time confirming compliance.
Lineage also supports internal confidence. Teams can troubleshoot issues faster, validate analytics outputs, and respond to regulatory questions without delay. What might otherwise require weeks of reconstruction becomes a matter of retrieval.
Why Lineage Is a Strategic Advantage?
Organisations that invest in lineage early avoid compliance debt. As portfolios grow, regions expand, and partners multiply, lineage scales naturally within PV-IT frameworks.
Without this foundation, complexity amplifies risk. With it, complexity becomes manageable.
Conclusion
In modern pharmacovigilance, data integrity is no longer proven by accuracy alone. It is proven by traceability. Data lineage provides the evidence that safety information has been controlled, validated, and understood at every step.
At Fidelity Health Services, PV-IT architectures are designed to preserve lineage across systems, vendors, and lifecycle stages. This ensures that when regulators ask how a decision was made, the answer is visible within the system itself.
In an inspection environment defined by scrutiny, lineage turns questions into confirmation.
Explore related insights in Data Validation and Migration in Pharmacovigilance and Compliance and Inspection Readiness in Pharmacovigilance .